The Chemistry Duo That Changed Flame Retardancy Forever
For much of the twentieth century, flame retardancy relied on a single dominant approach: halogens. Chlorine- and bromine-based systems delivered effective fire suppression and helped plastics meet safety standards quickly. But as material science evolved and as the environmental and health impacts of halogenated systems became clearer, the industry began searching for a smarter solution.
That search led to a powerful partnership in chemistry: phosphorus and nitrogen.
Together, these two elements reshaped how flame retardancy works, offering a cleaner, safer and more sustainable pathway for modern polymers.
Why Traditional Flame Retardants Reached Their Limits

Halogen-based flame retardants work primarily by interfering with the combustion process in the gas phase. While effective at stopping flames, they often generate dense smoke, toxic gases and corrosive by-products during a fire. In enclosed environments, these secondary effects pose serious risks to both people and equipment.
As global regulations tightened and industries adopted stricter fire-safety and environmental standards, manufacturers faced growing pressure to move away from halogens – without sacrificing performance.
The challenge was clear:
How do you maintain fire resistance while reducing toxicity and smoke?
The answer emerged through condensed-phase chemistry rather than gas-phase suppression. These limitations accelerated the industry’s transition toward antimony-free flame retardant systems, which prioritize lower toxicity, cleaner combustion, and long-term regulatory acceptance.
Understanding the Phosphorus–Nitrogen Synergy
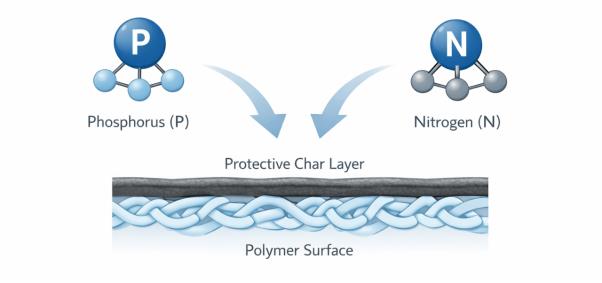
Individually, phosphorus and nitrogen each play an important role in flame retardancy. Together, they create a system that is far more effective than either element alone.
What Phosphorus Contributes
Phosphorus-based compounds promote char formation when exposed to heat. Instead of allowing the polymer to decompose and fuel the flame, phosphorus encourages the material to form a carbon-rich protective layer.
This char:
- Insulates the underlying material
- Slows heat transfer
- Reduces fuel availability
- Limits flame spread
What Nitrogen Adds
Nitrogen compounds support the process by:
- Releasing inert gases that dilute combustible vapours
- Enhancing intumescence (controlled swelling of the char layer)
- Improving the thermal stability of the flame-retardant system
The result is a stable, expanded char barrier that protects the polymer rather than reacting aggressively in the flame zone.
This cooperative mechanism is the foundation of phosphorus-nitrogen based flame retardants for plastics.
How Phosphorus–Nitrogen Systems Work During a Fire
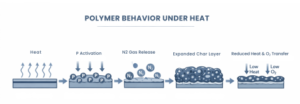
When a polymer containing a phosphorus–nitrogen flame retardant system is exposed to heat, the response follows a controlled sequence:
- Heat activationtriggers chemical reactions within the additive system
- Char-forming reactionsbegin at the polymer surface
- Nitrogen-driven expansioncreates an insulating intumescent layer
- Oxygen access is restricted, slowing combustion
- Smoke and toxic emissions are reduced
Unlike halogenated systems that suppress flames aggressively, phosphorus–nitrogen systems focus on preventing fire growth at the material level.
This approach aligns far better with modern safety expectations.
This condensed-phase behaviour is a defining characteristic of modern non-halogen flame retardant mechanisms, which focus on preventing fire growth rather than suppressing flames aggressively.
Why This Chemistry Changed the Industry
The adoption of phosphorus–nitrogen systems marked a turning point in flame retardancy. Manufacturers no longer had to choose between performance and responsibility.
Key advantages include:
- Halogen-free formulation
- Lower smoke density
- Reduced toxicity and corrosive gases
- Compatibility with RoHS and REACH requirements
- Improved performance in enclosed and sensitive environments
Because these systems work primarily in the condensed phase, they are particularly well-suited for applications where human safety and equipment protection are critical.
Applications Where Phosphorus–Nitrogen Systems Excel
Phosphorus–nitrogen flame retardants are now widely used across multiple polymer families and industries:
Polymer Systems
- Polypropylene (PP)
- Polyamides (PA6, PA66)
- Polyesters (PBT, PET)
- Engineering plastic blends
Application Areas
- Automotive interior and under-the-hood components
- Electrical and electronic housings
- Wire and cable insulation systems
- Consumer appliances
- Industrial enclosures
In many of these applications, achieving UL94 V-0 ratings while maintaining mechanical integrity is a critical requirement – one that phosphorus–nitrogen systems are well-positioned to meet.
The Role of Formulation Design
While phosphorus–nitrogen chemistry offers a strong foundation, performance still depends on how the system is formulated. Additive selection, loading levels, dispersion quality and compatibility with the base polymer all influence final results.
In practice, these systems are often combined with:
- Impact modifiers to maintain toughness
- Processing stabilizers for thermal control
- Synergists to fine-tune fire performance
This integrated approach allows compounders to tailor solutions for specific polymers, processing methods and end-use conditions.
Why This Chemistry Still Matters Today
Even as new flame-retardant technologies continue to emerge, phosphorus–nitrogen systems remain central to halogen-free design strategies. Their effectiveness, regulatory acceptance and adaptability make them a cornerstone of modern polymer safety.
More importantly, they represent a broader shift in material science – from reactive suppression to preventive protection.
Looking Ahead
The future of flame retardancy lies in systems that work with materials, not against them. Phosphorus–nitrogen chemistry proved that safer alternatives can also be high-performing, scalable and commercially viable.
As industries continue moving toward sustainable and responsible materials, this chemistry duo will remain a defining force in how fire safety is engineered into plastics.
Similar chemistry principles also support phosphorus–nitrogen systems in LSZH cable compounds, where smoke suppression and clean combustion are critical.
Designing fire-safe polymers today requires more than meeting minimum standards.
Explore halogen-free flame-retardant solutions built on proven phosphorus–nitrogen chemistry – engineered for performance, compliance and long-term reliability.